
Your Health Unlocked Episodes
041: Menopause Must-Knows in 2024 with the National Menopause Foundation
March 21, 2024
---
Consumer Health Info, Health Info
Publication Date: February 02, 2023
By: NWHN Staff

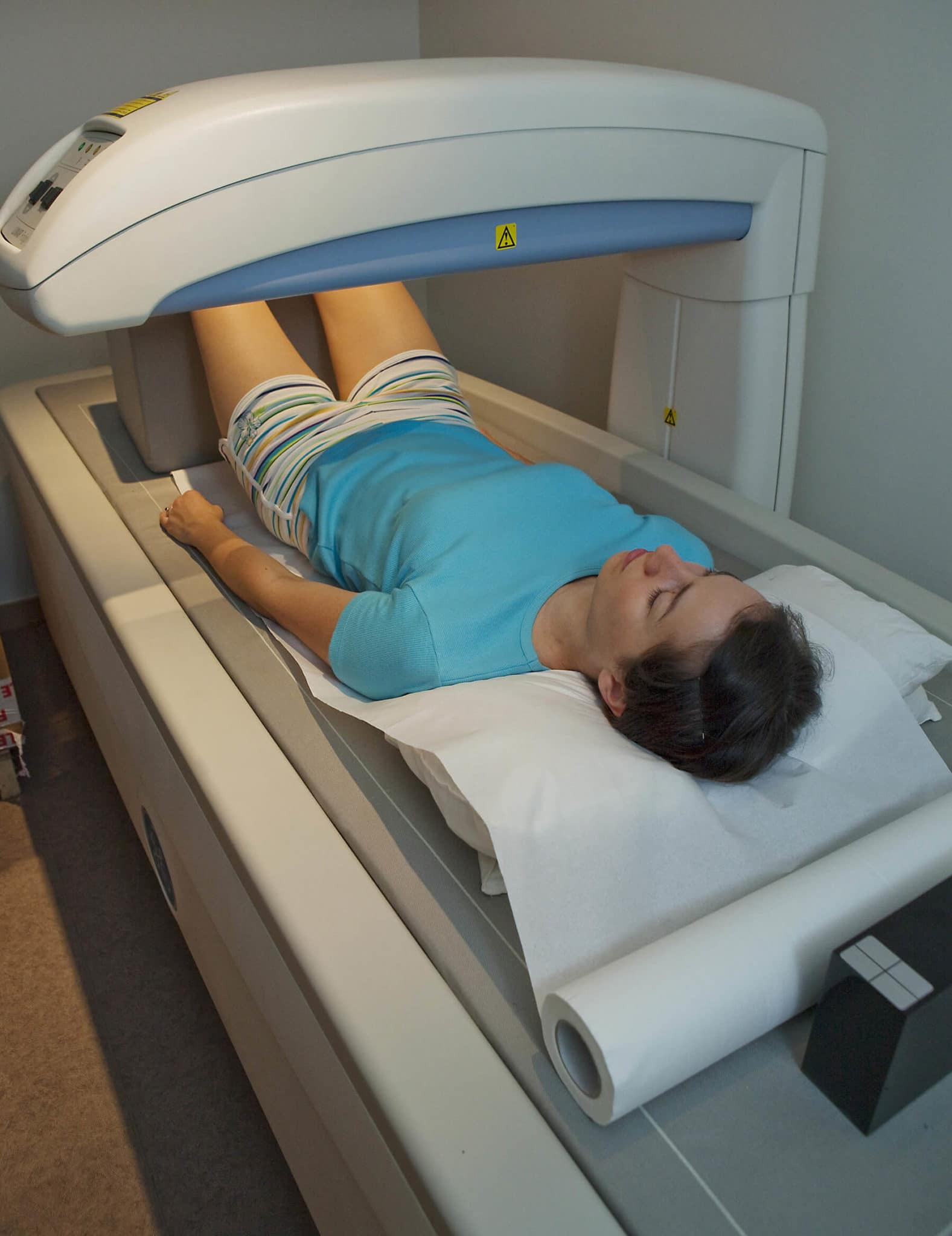 There is currently only one method used routinely to screen for osteoporosis, the DEXA test (short for dual-energy x-ray absorptiometry). The DEXA test attempts to predict which women are at risk of bone fracture based on an assessment of their bone density. But there’s a fundamental problem with this method – bone density is not necessarily the best predictor of future fracture. Many women with high bone density still fracture, and some women with low bone density may not need to be concerned.
There is currently only one method used routinely to screen for osteoporosis, the DEXA test (short for dual-energy x-ray absorptiometry). The DEXA test attempts to predict which women are at risk of bone fracture based on an assessment of their bone density. But there’s a fundamental problem with this method – bone density is not necessarily the best predictor of future fracture. Many women with high bone density still fracture, and some women with low bone density may not need to be concerned.
Not surprisingly, a screening test based on a flawed methodology has had some disappointing outcomes. Some studies show it is worse than or no more effective than simply assessing clinical risk factors, which can be done without expensive testing. DEXA has been shown to over-estimate how many women are at risk of fracturing. These “false positive” results lead many women to take drugs with serious risks and side effects despite being at low risk for fractures. The test even created a whole new classification called osteopenia, a term that described bone density loss but is not a condition or disease, that led to rampant over-prescription of osteoporosis drugs.
The goal of any screening technique should be to identify and treat women who are at high risk of fracture without exposing women who are at low risk of fracture to potentially dangerous drugs. We believe DEXA, when used alone, fails this goal. For these reasons, the National Women’s Health Network supports attempts to develop more precise and useful screening techniques.
This consumer health information will explore the developments in alternative screening technologies that could replace or improve DEXA. There have also been attempts to combine the bone density assessment provided by DEXA with clinical risk factors to produce a more accurate measurement to quantify risk. These algorithms have great implications for treatment decisions and will be presented in this content as well.
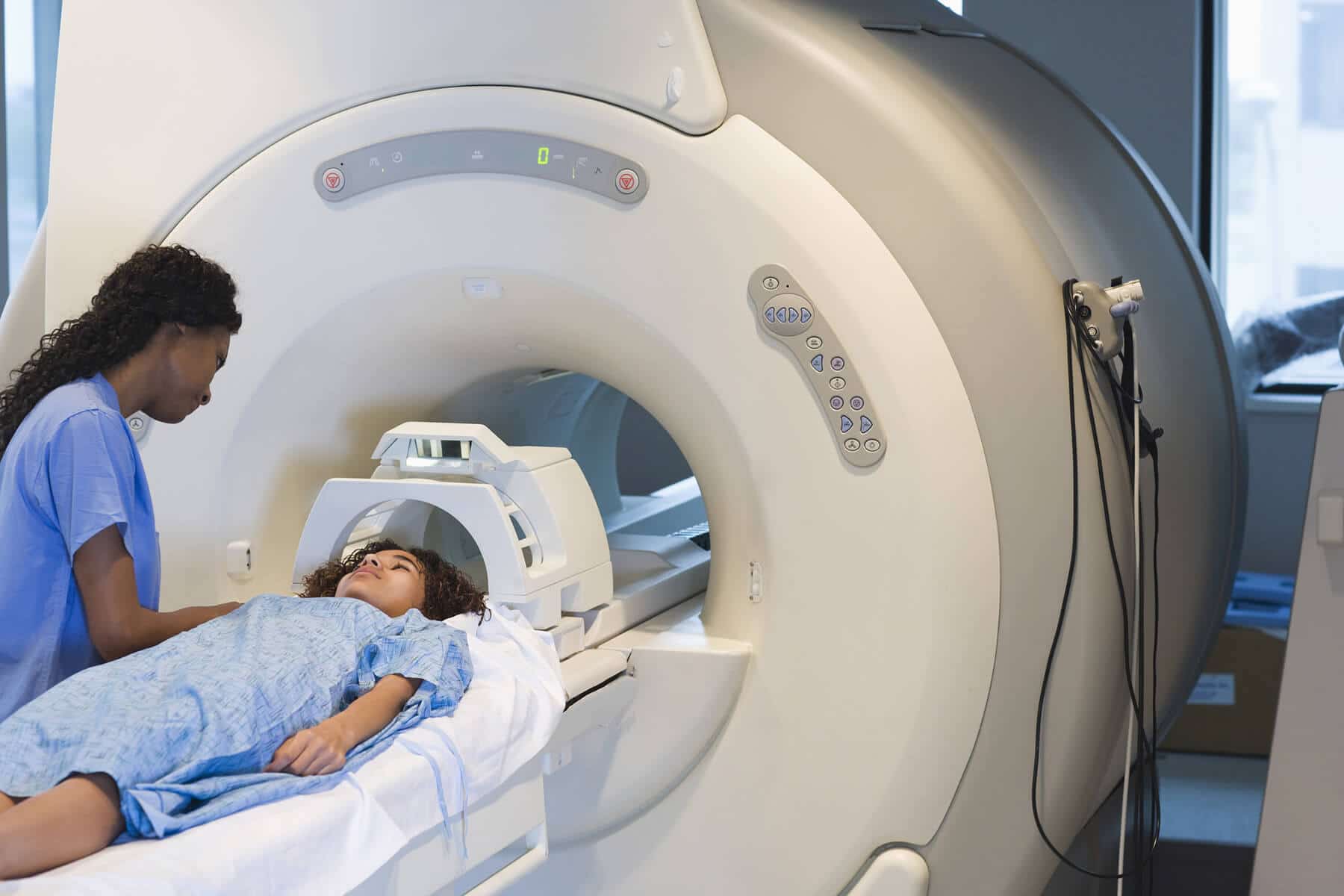 There is promising evidence that high-resolution magnetic resonance imaging (MRI) scans can offer a more precise form of screening than DEXA. The strength of an MRI machine is measured in Teslas (T) – the greater the number, the greater the resolution of an image. 3T MRIs are the most promising for osteoporosis screening because they offer a more detailed picture than standard 1.5 MRI scans can. These detailed 3TMRI images show microarchitectural details of bones, providing a much deeper insight into the bone strength than bone density alone. Bone texture, for example, is a biomarker that is known to predict fracture risk and can be detected by high-resolution MRI scanning techniques. 3TMRI is often used in research to give more precise assessment of whether an osteoporosis treatment actually strengthened bones. One major advantage of 3T MRI is that, like DEXA, it does not expose individuals to harmful radiation. As for its use in routine screening, one comparative study found that these 3T MRI scans could detect weak bones in individuals with prior fractures, even if they had normal bone density DEXA results.
There is promising evidence that high-resolution magnetic resonance imaging (MRI) scans can offer a more precise form of screening than DEXA. The strength of an MRI machine is measured in Teslas (T) – the greater the number, the greater the resolution of an image. 3T MRIs are the most promising for osteoporosis screening because they offer a more detailed picture than standard 1.5 MRI scans can. These detailed 3TMRI images show microarchitectural details of bones, providing a much deeper insight into the bone strength than bone density alone. Bone texture, for example, is a biomarker that is known to predict fracture risk and can be detected by high-resolution MRI scanning techniques. 3TMRI is often used in research to give more precise assessment of whether an osteoporosis treatment actually strengthened bones. One major advantage of 3T MRI is that, like DEXA, it does not expose individuals to harmful radiation. As for its use in routine screening, one comparative study found that these 3T MRI scans could detect weak bones in individuals with prior fractures, even if they had normal bone density DEXA results.
The results from this initial study are promising, but significant barriers would have to be overcome before 3T MRIs could become a routine part of osteoporosis screening. Additional studies, with many participants, taking place over a longer period of time, are needed to adequately determine whether 3T MRI scans can actually predict fractures. MRI scans require an experienced technician to look at the image and to interpret the results, making 3T MRI less convenient than DEXA, which produces a simple numerical output. 3T MRIs are also more expensive than DEXA scans and other experimental screening techniques like quantitative computated tomography (QCT). Though 3T MRIs are not yet ready to be used for routine screening, the NWHN believes this technology shows great promise. Researchers should continue to explore the accuracy and feasibility of using 3T MRI as a routine osteoporosis screening tool.
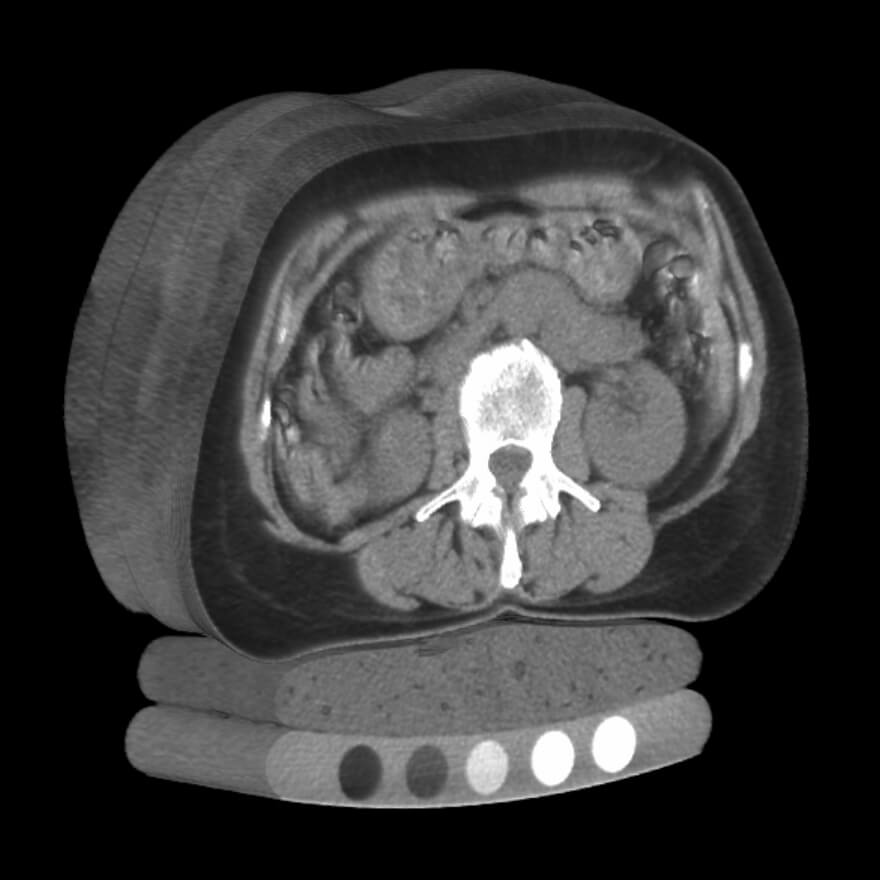
High resolution, three dimensional quantitative computated tomography (QCT) can offer additional insight into bone strength than DEXA depending on where the measurement is taken. QCT of the hip simply offers another measurement of bone mineral density. However, when QCT scans are used to view the spine, it provides insight into the deep spongy bone that DEXA bone mineral density testing cannot measure. This spongy bone, called trabecular bone, has a high metabolic rate and can show deterioration in bone strength before traditional bone mineral density changes.
QCT, just like 3T MRI, is regularly studied in research settings and it shows promise as a screening method. Studies have shown, similar to 3T MRIs, QCT is highly effective at identifying bones that are weak, even when bone mineral density results are normal. However, QCT exposes women to higher doses of radiation than DEXA or MRI. This has curbed enthusiasm to invest in QCT as technique for routine screening. This test is still an important research tool, where it is used to identify microarchitectural bone characteristics associated with fractures and analyze trends in fracture recovery. Despite its value to researchers, it remains to be seen how this technology could be applied in a clinical setting.
Because DEXA scanning has become a part of routine care, it would be hard to uproot and replace it entirely. Despite DEXA’s flaws, it provides a number that quantifies risk and helps simplify diagnosis and treatment decisions. For better or worse, it is easier for providers to make recommendations based on clear-cut quantitative benchmarks. Some attempts are being made to help produce quantitative tools that work alongside DEXA screening, but include other risk factors, offering a more holistic assessment of fracture risk.
The FRAXTM algorithm is one of these tools. A provider inputs a person’s DEXA score and risk factors, and FRAXTM calculates the person’s statistical probability of fracture within the next ten years. The model differentiates between different populations based on geography and ethnicity, where data is available. FRAXTM is not the only tool that tries to quantify risk of osteoporotic fracture, but it is the most widely used and studied to date. Several studies indicate that FRAXTM is a useful tool for making treatment decisions and that it is at least as good, perhaps better, at accurately predicting fracture risk than DEXA bone mineral density results alone.
The NWHN applauds the shift from an exclusive focus on DEXA results. However, the details of the FRAXTM algorithm and underlying data haven’t been disclosed to the public. We are particularly concerned with how heavily DEXA scores might be weighed compared to other individual risk factors. The secrecy prevents independent organizations like the NWHN from truly studying and analyzing these tools. The World Health Organization (WHO) has stated it is unable to assess the tool and make a recommendation for use (or not) until the algorithm and supporting data is public information. The NWHN encourages women to approach this tool with reservation and caution.
Once a procedure becomes routine, it is difficult to replace. However, the NWHN is committed to a vision of osteoporosis care where all women who need it can receive effective screening in order to prevent unnecessary fractures through both medication and non-pharmacologic interventions.
At present, it is hard to imagine screening techniques like 3TMRI or QCT moving beyond medical research and becoming a standard part of osteoporosis screening at this point in time. However, FRAXTM and other algorithms are already poised to make a lasting impact on how we screen and treat osteoporosis. We will continue to raise our concerns over the secrecy of these algorithms and push for better transparency. You can trust that the National Women’s Health Network will be watching these technologies closely and keep pushing for women to get the medical care they need.
